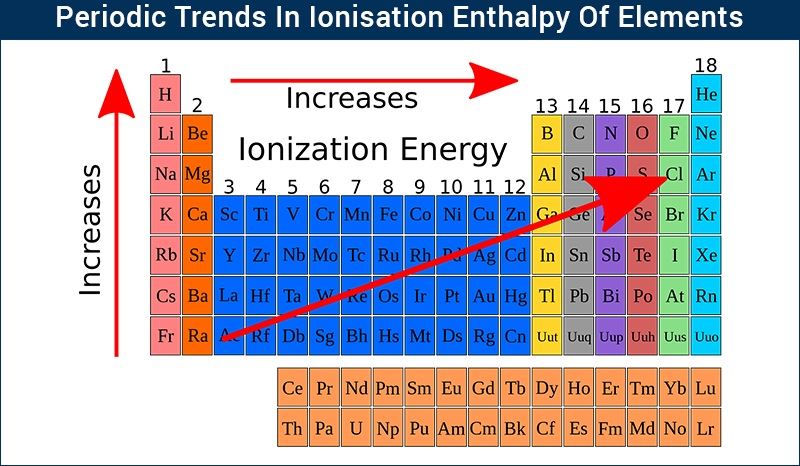
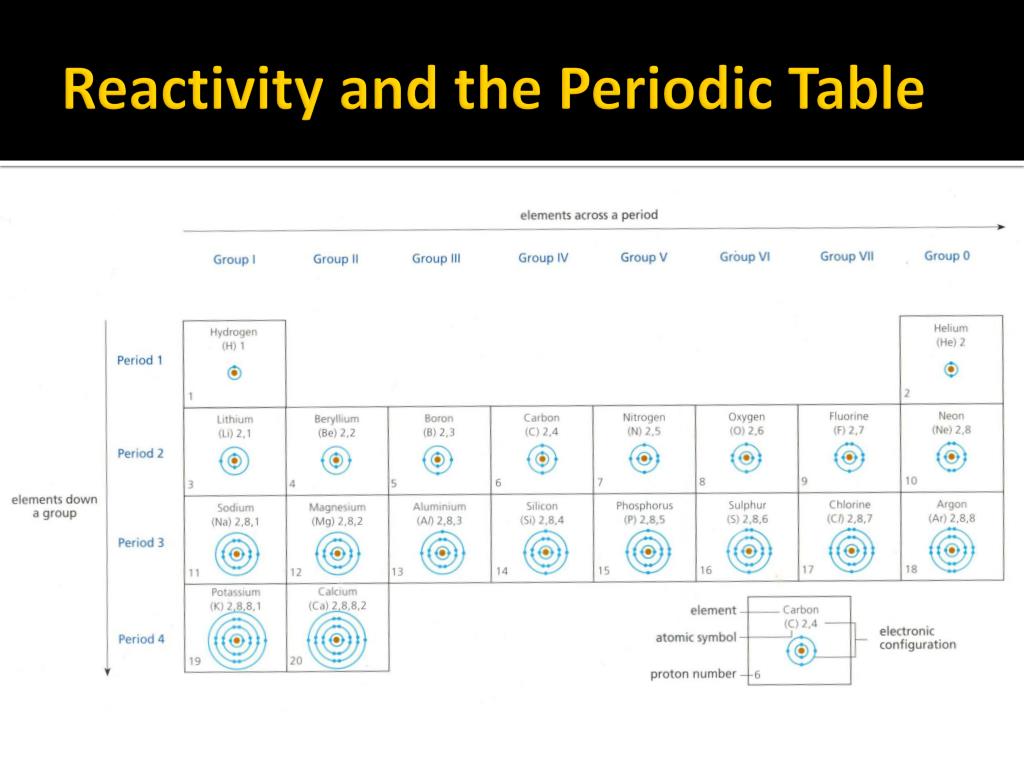
 Among transition metals (3d series), the change in atomic radii is much smaller as compared to those of representative elements across the period.This is because the elements present at left extreme of a period have the lowest ionization enthalpy and can lose an electron to form a cation and that present at the right extreme have the highest electron affinity and can gain an electron to form an anion. Chemical reactivity is highest at the two extremes of a period and is lowest at the center.Non availability of d-orbitals for bonding.Element of second period are known as bridge elements. These pairs of element have almost identical ionic radii and polarizing power (i.e. Anomalous properties of second period elements – diagonal relationshipĬertain elements in the 2 nd period show similarity with their diagonal elements in 3 rd period. The stability of the +1 oxidation state increases in the following sequence:Ģ. As an example in group 13 the +1 oxidation state of Tl is the most stable and Tl3+ compounds are comparatively rare.The term inert pair effect is often used in relation to the increasing stability of oxidation states that are 2 less than the group valency for the heavier elements of groups 13, 14, 15 and 16. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
